Explore pharma jobs for freshers and experienced candidates at Lambda Therapeutic Research, apply online for Clinical Research Associate role today.
Introduction
If you are looking to advance your career in clinical research and business intelligence within the pharma sector, this role offers a unique blend of data analysis and clinical trial exposure. The Clinical Research Associate I at Lambda Therapeutic Research Ltd. is an excellent opportunity for experienced professionals who want to work in a global CRO environment and contribute to strategic research and business development activities.
Job Overview
| Details | Information |
|---|---|
| Job Title | Clinical Research Associate I |
| Company Name | Lambda Therapeutic Research Ltd. |
| Location | Ahmedabad, India |
| Employment Type | Full-Time |
| Department | Clinical Research / Business Development |
| Experience Required | Around 5 Years |
| Education | B.Pharm / M.Pharm / MSc (Life Sciences) |
| Salary | ₹5,00,000 – ₹8,00,000 per annum |
Company Overview
Lambda Therapeutic Research Ltd. is a globally recognized Contract Research Organization (CRO) headquartered in Ahmedabad. With operations across India, USA, Canada, UK, Spain, and Poland, the company delivers end-to-end clinical research services to pharmaceutical and biotech clients.
With a workforce of over 1,200 professionals and advanced digital infrastructure, Lambda ensures high-quality data integrity, regulatory compliance, and efficient project execution. The organization has successfully cleared numerous international inspections by authorities like USFDA, EMA, and MHRA, reinforcing its strong global reputation.
Job Location & Employment Type
- Location: Ahmedabad, Gujarat
- Employment Type: Full-time
- Work Mode: On-site
Open Positions / Department Details
- Position: Clinical Research Associate I
- Department: Clinical Research / Business Development Support
- Function: Data analysis, lead generation, and regulatory intelligence
Key Roles & Responsibilities
The selected candidate will be responsible for:
- Researching clinical trial registries, SEC filings, and ANDA databases
- Identifying potential sponsors and business opportunities
- Building and maintaining lead databases using Excel
- Performing data mining and analysis for pharma pipelines and clinical trials
- Tracking regulatory approvals and company developments
- Preparing reports, dashboards, and actionable insights
- Using AI-based tools like Apollo.ai for outreach and prospecting
- Supporting Business Development (BD) and Clinical Trial Management teams
- Improving targeting strategies through data-driven insights
Eligibility Criteria
Education
- B.Pharm / M.Pharm
- MSc in Life Sciences or equivalent
Experience
- Around 5 years of experience in clinical research
- Experience in clinical trials, CRO environment, or business development preferred
- Exposure to regulatory databases and market research is beneficial
Skills Required
- Strong Microsoft Excel skills (pivot tables, filtering, analytics)
- Knowledge of clinical trial databases and regulatory systems
- Analytical thinking and data interpretation ability
- Familiarity with AI tools (e.g., Apollo.ai, CRM platforms)
- Excellent communication and coordination skills
- Strong organizational and documentation ability
Salary & Benefits
- CTC Range: ₹5,00,000 – ₹8,00,000 per annum
Additional Benefits
- Work in a globally established CRO
- Exposure to international regulatory frameworks
- Opportunity to work on data-driven pharma strategies
- Career growth in clinical research and business intelligence
- Learning exposure to AI tools and automation in pharma
Selection Process
The recruitment process typically includes:
- Application screening
- Technical interview (clinical research + data analysis skills)
- HR interview
- Final selection
How to Apply
Candidates interested in the Clinical Research Associate I at Lambda Therapeutic Research Ltd. can apply through the official company careers page (online application process).
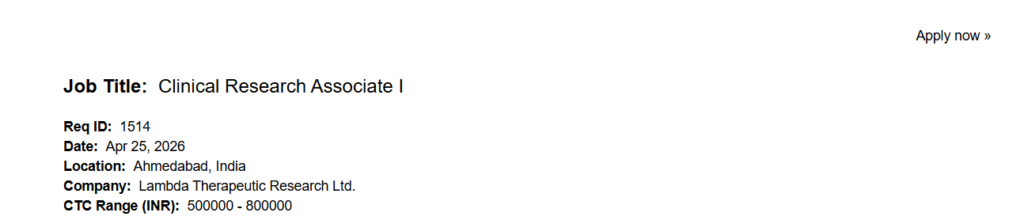
Important Dates
- Last Date to Apply: Not specified
Why Apply for This Job?
This role is ideal for professionals who want to combine clinical research expertise with data analytics and business strategy:
- Opportunity to work with a reputed global CRO
- Exposure to clinical trials, regulatory data, and pharma pipelines
- Develop expertise in business development and market intelligence
- Hands-on experience with AI-based prospecting tools
- Strong long-term career growth in pharma analytics and CRO sector
FAQs
1. What does a Clinical Research Associate I do at Lambda Therapeutic Research Ltd.?
The role involves analyzing clinical trial data, generating business leads, and supporting BD teams with insights.
2. What qualifications are required for this position?
Candidates need B.Pharm, M.Pharm, or MSc in Life Sciences.
3. How much experience is needed for this job?
Around 5 years of experience in clinical research is required.
4. What salary is offered for this role?
The salary ranges between ₹5 LPA and ₹8 LPA.
5. How can I apply for this job?
You can apply online through the official Lambda Therapeutic Research careers portal.
Conclusion
The Clinical Research Associate I at Lambda Therapeutic Research Ltd. is a strong career opportunity for experienced professionals in clinical research and pharma data analytics. With global exposure, competitive salary, and skill development opportunities, this role can significantly enhance your professional growth. Apply soon to take the next step in your pharma career.
Disclaimer
LatestPharmaJobs.in is an independent job information platform. We do not provide jobs or recruitment and are not affiliated with any company.

