Latest pharma jobs for freshers and experienced candidates apply online for Ipca Laboratories QA QC walk-in interview eligibility salary details now.
Introduction
If you have experience in API pharmaceutical manufacturing and want to advance your career, the Quality Assurance & Quality Control Ipca Laboratories hiring drive is a great opportunity. This walk-in interview is open for experienced professionals across QA and QC roles, offering exposure to regulated environments and global compliance standards.
Job Overview
| Field | Details |
|---|---|
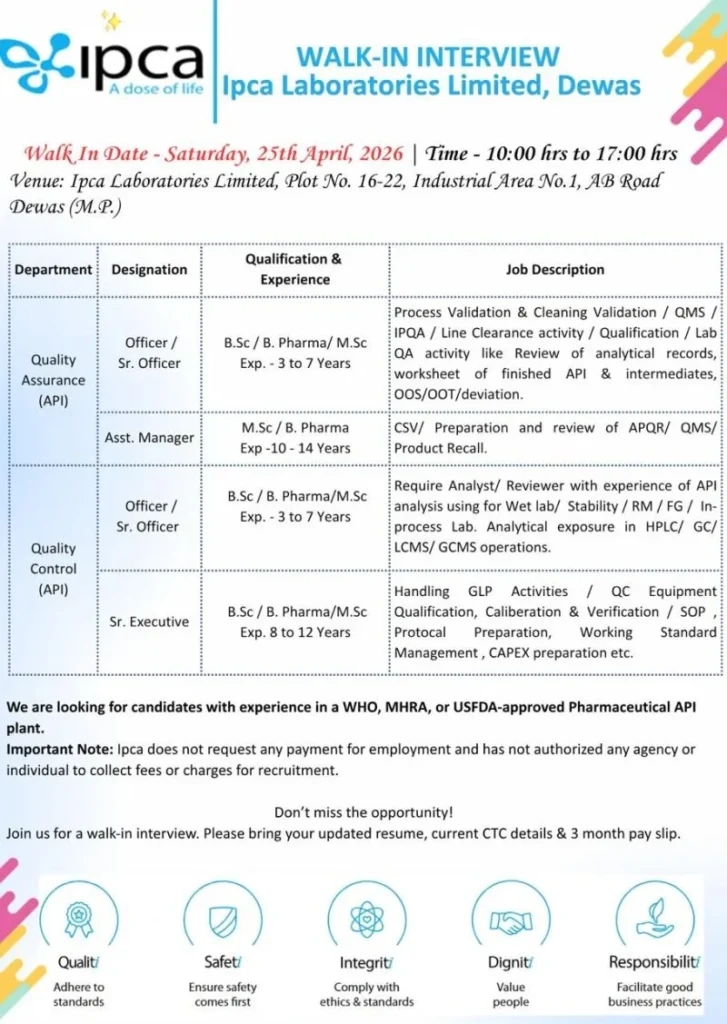
| Job Title | QA & QC (Multiple Roles) |
| Company Name | Ipca Laboratories |
| Location | Dewas, Madhya Pradesh |
| Employment Type | Full-Time |
| Department | Quality Assurance / Quality Control |
| Experience Required | 3–14 Years |
| Education | B.Sc / B.Pharm / M.Sc |
| Salary | ₹3 – ₹10 LPA |
Company Overview
Ipca Laboratories is a well-known pharmaceutical company with strong expertise in API manufacturing and global regulatory compliance. The company operates in highly regulated markets and is recognized for maintaining high-quality standards across its manufacturing units.
Working at Ipca provides professionals with exposure to international guidelines such as WHO, USFDA, and MHRA, making it a valuable career move for experienced candidates.
Job Location & Employment Type
- Location: Dewas, Madhya Pradesh
- Employment Type: Full-Time (On-site)
This role is based at Ipca’s API manufacturing facility, offering hands-on experience in regulated production and quality systems.
Open Positions / Department Details
Quality Assurance (API)
- Officer / Sr. Officer (3–7 years)
- Assistant Manager (10–14 years)
Quality Control (API)
- Officer / Sr. Officer (3–7 years)
- Sr. Executive (8–12 years)
These roles are part of the API Quality Department, responsible for ensuring product quality, compliance, and regulatory adherence.
Key Roles & Responsibilities
Quality Assurance (QA)
- Perform process validation and cleaning validation
- Manage QMS, IPQA, and line clearance activities
- Handle qualification and laboratory QA processes
- Review analytical records and API documentation
- Manage OOS, OOT, and deviation investigations
- Prepare APQR reports and support product recall processes
- Work on CSV (Computer System Validation) activities
Quality Control (QC)
- Conduct API analysis (RM, in-process, FG, and stability samples)
- Operate instruments like HPLC, GC, LCMS, and GCMS
- Ensure GLP compliance and proper documentation
- Perform calibration and qualification of equipment
- Prepare SOPs, protocols, and reports
- Support CAPEX and standardization processes
Eligibility Criteria
Education
- B.Sc / B.Pharm / M.Sc in relevant discipline
Experience
- Minimum 3 years to 14 years in API pharmaceutical industry
Skills Required
- Strong knowledge of GMP and regulatory guidelines
- Experience in WHO, MHRA, or USFDA approved plants preferred
- Hands-on experience in QA/QC processes
- Analytical thinking and problem-solving ability
- Attention to detail and documentation accuracy
Salary & Benefits
- Salary: ₹3 LPA – ₹10 LPA (depending on experience and role)
- Work in a globally regulated manufacturing environment
- Exposure to international quality standards
- Career advancement opportunities
- Stable and reputed organization
Selection Process
This is a walk-in interview, and selection includes:
- Document verification
- Technical interview
- HR discussion
- Final selection
How to Apply
Candidates must attend the walk-in interview at the following venue:
Venue:
Ipca Laboratories Limited
Plot No. 16-22, Industrial Area No.1
AB Road, Dewas, Madhya Pradesh
Documents to Carry:
- Updated resume
- Current CTC details
- Last 3 months’ salary slips
Important Dates
- Walk-in Date: 25 April 2026 (Saturday)
- Time: 10:00 AM to 05:00 PM
Why Apply for This Job?
The Quality Assurance & Quality Control Ipca Laboratories opportunity is ideal for experienced pharma professionals:
- Work in a WHO, USFDA, and MHRA-compliant environment
- Gain experience in advanced QA/QC systems
- Strong career growth in API pharmaceutical industry
- Opportunity to work with a reputed pharma company
- Competitive salary based on experience
FAQs
1. What is the focus keyword for this job?
The focus keyword is Quality Assurance Quality Control Ipca Laboratories.
2. Can freshers apply for this role?
No, this role requires a minimum of 3 years of experience.
3. What is the salary range?
The salary ranges from ₹3 LPA to ₹10 LPA depending on role and experience.
4. Is this an online application?
No, this is a walk-in interview opportunity.
5. What experience is preferred?
Experience in API pharma and regulated plants (WHO/USFDA/MHRA) is preferred.
Conclusion
The Quality Assurance & Quality Control Ipca Laboratories walk-in drive is a valuable opportunity for experienced professionals in API pharma. With strong exposure to global regulatory standards and excellent career growth prospects, this role is ideal for candidates looking to advance in QA/QC domains. Attend the interview on time and take the next step in your pharma career.
Disclaimer
LatestPharmaJobs.in is an independent job information platform. We do not provide jobs or recruitment and are not affiliated with any company.

