Apply pharma jobs for freshers and experienced professionals. Apply online for Senior Manager Production Sun Pharma role with eligibility details now.
Introduction
Looking to step into a leadership role in pharmaceutical manufacturing? The Senior Manager Production (Parenteral/Injectables) at Sun Pharma Medicare Ltd offers an excellent opportunity for experienced professionals in sterile manufacturing. This role is ideal for candidates with strong expertise in injectables, regulatory compliance, and team management, especially those with exposure to USFDA audits and global standards.
Job Overview
| Field | Details |
|---|---|
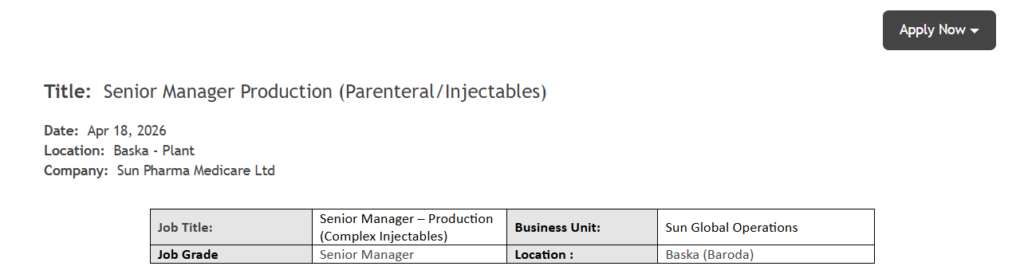
| Job Title | Senior Manager – Production (Complex Injectables) |
| Company Name | Sun Pharma Medicare Ltd |
| Location | Baska (Baroda), Gujarat |
| Employment Type | Full-Time |
| Department | Sun Global Operations |
| Experience Required | Minimum 14 Years |
| Education | M.Sc / B.Pharm / M.Pharm |
| Salary | As per company norms |
Company Overview
Sun Pharma Medicare Ltd is part of the globally recognized Sun Pharma group, known for its excellence in manufacturing high-quality pharmaceutical products. The company specializes in advanced dosage forms, including complex injectables, and follows stringent global regulatory standards. It provides a dynamic work environment where experienced professionals can lead high-impact projects and contribute to global healthcare.
Job Location & Employment Type
- Location: Baska (Vadodara), Gujarat
- Work Mode: On-site (Manufacturing Plant)
- Employment Type: Full-Time
This role is based at a manufacturing facility, requiring hands-on involvement in production operations and leadership.
Open Positions / Department Details
- Department: Sun Global Operations (Production)
- Role Level: Senior Manager
- Industry: Pharmaceutical Manufacturing
- Focus Area: Parenteral / Injectable Production
This is a senior leadership role responsible for managing sterile manufacturing processes and ensuring compliance with global regulatory standards.
Key Roles & Responsibilities
As a Senior Manager – Production (Injectables), your responsibilities will include:
- Monitoring and controlling manufacturing and filling line operations
- Implementing process improvements to enhance product quality and efficiency
- Reviewing and approving key production documents such as:
- Master Formula (MF)
- Batch Manufacturing Records (BMR)
- Bill of Materials (BOM)
- Managing SAP-related production activities
- Ensuring proper qualification, calibration, and maintenance of equipment
- Maintaining compliance with regulatory standards and operational procedures
- Driving strategic initiatives to improve manufacturing performance
- Conducting training programs to enhance team productivity and skills
- Preparing for and handling internal and external audits (including USFDA audits)
- Ensuring validation activities are completed as per established protocols
- Maintaining audit readiness at all times
Eligibility Criteria
Education
- M.Sc, B.Pharm, or M.Pharm from a recognized institution
Experience
- Minimum 14 years of experience in injectable manufacturing
- Mandatory experience in handling USFDA audits
Skills Required
- Strong expertise in sterile and injectable production processes
- Knowledge of GMP, regulatory compliance, and validation procedures
- Experience in handling audits and inspections
- Leadership and team management skills
- Strong documentation and review capabilities
- Problem-solving and decision-making abilities
Salary & Benefits
- Salary: As per company norms
- Leadership role in a reputed pharmaceutical organization
- Exposure to global manufacturing standards
- Opportunities for career advancement
- Training and development programs
- Collaborative and growth-focused work environment
Selection Process
The recruitment process generally includes:
- Online application submission
- Resume shortlisting
- Technical interview (Production & Injectables expertise)
- Managerial/Leadership round
- HR discussion
- Final selection
Candidates should prepare thoroughly on injectable manufacturing, GMP compliance, and audit handling.
How to Apply
Eligible candidates can apply through the online application process via the official company career portal.
Steps to apply:
- Visit the official careers page
- Search for “Senior Manager Production (Injectables)”
- Register or log in
- Submit your updated resume
- Complete the application form
Important Dates
- Application Deadline: Not specified (Apply early recommended)
Why Apply for This Job?
The Senior Manager Production at Sun Pharma Medicare Ltd role offers exceptional career benefits:
- Opportunity to lead complex injectable manufacturing operations
- Work with a globally recognized pharmaceutical leader
- Gain exposure to USFDA and international regulatory standards
- Enhance leadership and strategic decision-making skills
- Contribute directly to high-quality pharmaceutical production
This role is ideal for experienced professionals aiming for higher leadership positions such as Plant Head or Director of Manufacturing.
FAQs
1. What experience is required for this role?
A minimum of 14 years of experience in injectable manufacturing is required.
2. Is USFDA audit experience mandatory?
Yes, candidates must have experience handling USFDA audits.
3. What qualifications are needed?
Candidates should have M.Sc, B.Pharm, or M.Pharm degrees.
4. Where is the job location?
The job is located in Baska (Vadodara), Gujarat.
5. What is the salary offered?
Salary is not disclosed and will be as per company norms.
Conclusion
The Senior Manager Production (Parenteral/Injectables) at Sun Pharma Medicare Ltd 2026 is a high-level opportunity for experienced professionals in pharmaceutical manufacturing. With leadership responsibilities, global exposure, and strong career growth potential, this role is ideal for those looking to advance in the injectables domain. Apply early to take the next step in your career.
Disclaimer
LatestPharmaJobs.in is an independent job information platform. We do not provide jobs or recruitment and are not affiliated with any company.

